  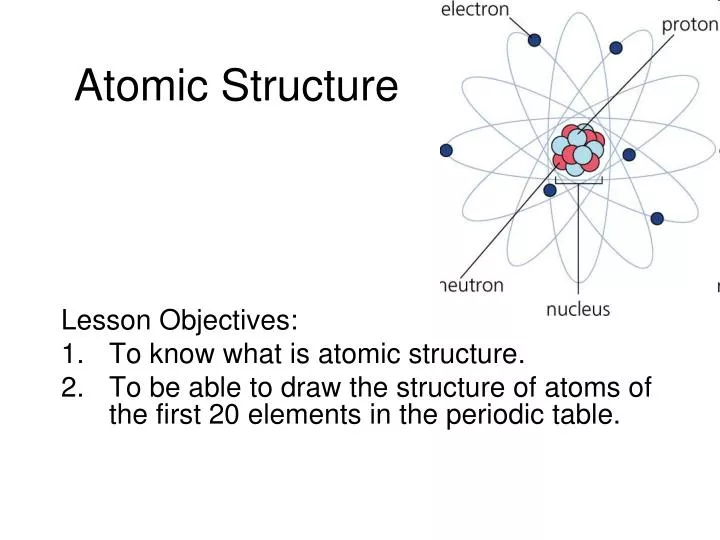
The crystal is cut parallel to a set of its -planes and bombardment is at normal incidence. it" of scattering of a homogeneous beam of electrons of adjustable speed incident upon a single crystal of nickel has been measured as a function of direction.Germer experiment Electron scattering peak at 500 1929 Nobel Prize for de Broglie PHYSICAL REVIEW DIFFRACTION OF ELECTRONS BY A CRYSTAL OF NICKEL BY C. 30, 6 1924 de Broglie's hypothesis 0 Nickel stal 1927 Davisson. Electron gun hot filament to Mrelease electrons +54 V Accelerating o electrode Theory -1.67Åfor54V Experiment Pathlength difference d sine 1.65Å for constructive interference Not bad for a three ear old idea! Nickel lattice spacing d = 2,15Å Second Series December, 1927 THE Vol. They discovered the wave nature of electron by getting a diffraction pattern from a Ni crystal by bombarding a beam of electron. Please CLICK HERE to have a look around and don’t forget to follow if you like what you see.Īlso, please visit my blog Teach With Fergy for my thoughts on teaching, learning and everything in between.Experimental proof of de Broglie hypothesis Davisson-Germer experiment (1923-1927) confirms the de Broglie hypothesis (wave-particle duality). I also have a range of assignments and lessons/units from other grade levels at my store. powerpoint-atomicstrucutureib.ppt 1986 kb ppt 2-atomicstructureworksheets-answers-grade11ib.docx 1917 kb docx 3-atomicstructureworksheets. This also leaves more time for discussion and activities. I have found this to be the most effective means of keeping my students engaged and active without having them write everything out. Isotopes atoms of the same element that have different atomic mass numbers due to different numbers of neutrons. These blanks are conveniently underlined and bolded on the teacher copy. The student version of the PowerPoint contains multiple blanks that need to be filled in throughout the lesson. CHAPTER 2 LESSON 1 The Nature of Matter READING TOOL Use Structure As you read. VSEPR Theory VSEPR Theory 4 Shapes of molecules 1. Dipoles) PowerPoint Presentation Space filling model Electron-Cloud model Water is asymmetrical Water is a bent molecule PowerPoint Presentation Making sense of the polar non-polar thing IONIC bonds. The student version of the presentation The diagram shows a model of a carbon atom, with an atomic number of 6. non-polar MOLECULES Polar molecules (a.k.a. The teacher version of the presentation Office and Google versions of the presentation 
silicon & oxygen make up 75 of earth’s crust 92. most abundant elements in universe hydrogen & helium 5. periodic table system for classifying elements 4. 
symbol of one or two letters represent each 3. forms of matter that contain only one type of atom 2. It safeguards you in case your department or school decides to go Google 100% and allows you as well as your students to access everything no matter where you are or what kind of equipment is being used. elements cannot be broken down into simpler form by ordinary chemical means 1. Having both options allows you to use either without worrying about formatting issues as all the work has already been done for you. Purchase -> Open -> Click -> Assign to your students Your resource is perfect for distance learning and integrates seamlessly with Google Classroom as I've included a link that will automatically create a copy of the resources and put them directly into your Google Drive with one click. The presentation is in PowerPoint and Google Slides while all documents contain Word and Google Doc versions. This lesson includes both Office and Google formats for ALL resources. These shells explain the whole of chemistry. Thevolumetheir orbits occupy determines how big the atom is. This package includes the lesson (student and teacher versions of the presentation), an auto-graded Exit Ticket to check student understanding, a Bill Nye worksheet (includes answer key), and a student lesson handout as a document. They’retiny, but they cover a lot of space. Furthermore, it explains Atomic Number, Mass Number, and Atomic Symbols. This lesson package introduces Atoms, Protons, Neutrons, and Electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
